|
We should remember that a molecule described as a resonance hybrid never possesses an electronic structure described by either resonance form. A double-headed arrow between Lewis structures indicates that they are resonance forms. The actual electronic structure of the molecule (the average of the resonance forms) is called a resonance hybrid of the individual resonance forms. We call the individual Lewis structures resonance forms. Part of this variety in organic chemistry stems from differences in electron behavior when elements other than carbon and hydrogen participate in molecular. The acetate anion has two resonance structures of the.  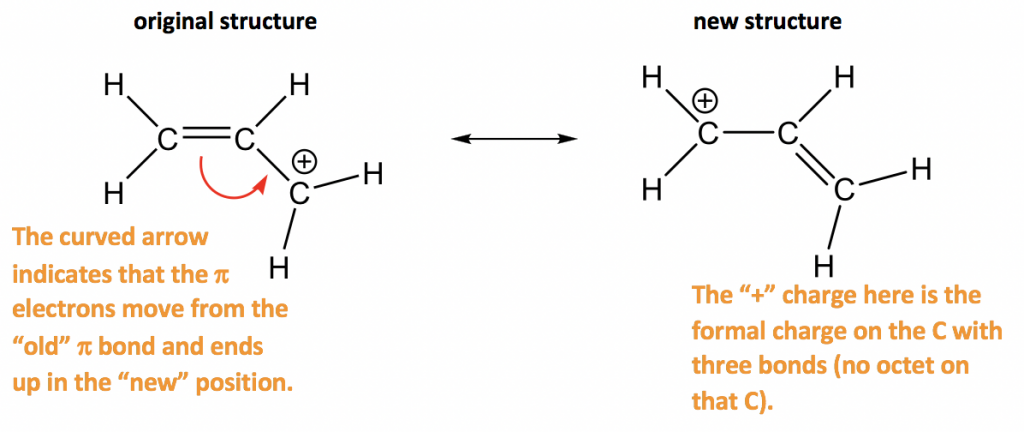
We as chemists draw so-called resonance structures to depict each possible snapshot of the molecule that contribute to the overall observed resonance hybrid structure. The actual distribution of electrons in each of the nitrogen-oxygen bonds in is the average of a double bond and a single bond. Resonance is a molecules way of spreading out its electron density, and that helps to minimize its ground-state energy. Instead, we use the concept of resonance: if two or more Lewis structures with the same arrangement of atoms can be written for a molecule or ion, the actual distribution of electrons is an average of that shown by the various Lewis structures. It is not possible to write a single Lewis structure for in which nitrogen has an octet and both bonds are equivalent. Experiments show, however, that both N–O bonds in have the same strength and length, and are identical in all other properties. A double bond between two atoms is shorter (and stronger) than a single bond between the same two atoms. Counting electrons: Resonance and acid-base chemistry Resonance structures: Resonance and acid-base chemistry Organic acid-base chemistry: Resonance and acid-base chemistry. What are Resonance Structures Resonance structures are the Lewis structure sets that describe the electron’s delocalization in a molecule or a polyatomic ion. Register free for online tutoring session to clear your doubts. 
In the VB theory case it is simply implied that the energy of the electron(s) involved in resonance is similar in the postulated geometries and that there exists a path allowing the electrons to shift between bonds involved in resonance. Learn about resonance structures topic of chemistry in details explained by subject experts on. If nitrite ions do indeed contain a single and a double bond, we would expect for the two bond lengths to be different. Basic understanding of basic high school or college chemistry assumed (although there is some review). Resonance is a phenomenon in which energy is exchanged between tuned systems (such as radio antennae). Double-headed arrows link the different resonance structures of a compound: Figure 2.4.1). The position of the atoms is the same in the various resonance structures of a compound, but the position of the electrons is different. \) may actually be drawn in two different ways, distinguished by the locations of the N-O and N=O bonds: Equivalent Lewis dot structures, such as those of ozone, are called resonance structures.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
